Product Description
The Gene-Foci cloning fast screening kit is designed as an initial screen for the positive clone after ligating the target gene into a vector and E.coli transformation. The kit can identify positive clones by size difference without mini-prep DNA extraction from transformants.
Kit content:
2 X 500 µl Fast Screen buffer.
Features:
Initial identification of the positive clones containing the gene insert without mini-prep DNA extraction, enzyme digestion. Saves time and labor.
Just 20 µl of liquid culture or half of a medium-sized colony is sufficient for screening.
Bright-colored gel loading dye included, easy gel loading after incubation.
Storage:
Store at room temperature.
Protocol:
Before Start:
- Warm up a water bath or heat block to 70°C
- Warm up the fast screen buffer to >37°C to let the precipitate get back into solution.
- Perform enzyme digestion, ligation, and E.coli competent cell transformation. Also transform the empty vector into E.coli as a control.
- Pick 10~30 transformants, set up 5 ml culture in liquid LB media plus the appropriate antibiotics, also set up a 5 ml culture for the empty vector as a control. Culture in a 37°C shaker overnight.
Alternatively, if the colonies are >2mm in diameter, you can skip the liquid culture step, directly pick half of a colony with a sterilized tooth pick, transfer to a 1.5 ml tube, and precede to step 4. - Pellet 20~100 µl overnight E.coli culture at 13,000 rpm for 30 seconds, carefully aspirate or pipette off the supernatant without disturbing the cell pellet.
- Resuspend the cell pellet in 20µl fast screen buffer by gently pipetting up and down, avoiding bubbles.
- Heat at 70°C for 10 min.
- Cool down to room temperature and run out the entire 20 µ on a 0.7% – 1% agarose gel.
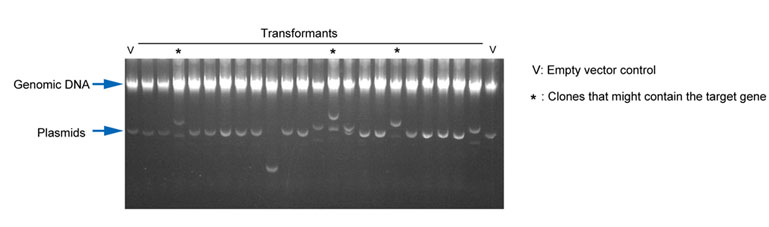
- Pick the clones that migrate slower than the vector control, mini-prep to extract DNA from the original 5 ml culture, then further validate by enzyme digestion and/or DNA sequencing.
Fig1. An example of selecting positive clones from an experiment attempting to clone a target gene into a very difficult to handle Lentiviral plasmid using Fast Screen buffer.
Helpful hints:
- Make sure gel running buffer is level with the surface of the gel, and not above the level of the gel.
- Remove running buffer from the wells of the gel using a pipette to avoid overflow when loading samples.
- Pipette lysate up and down several times before loading onto gel to reduce viscosity.